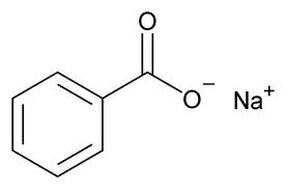
Sodium Benzoate Molecule
Molecular Formula: NaC7H5O2 | Molar Mass: 144.11 g/mol | Melting Point: 300 °C | Density: 1.50 g/cm³
Sodium benzoate is a white, crystalline powder that is a result of the reaction of benzoic acid with sodium hydroxide. In large concentrations, it holds an unpleasant, saline, astringent taste. The optimum pH of this chemical is between 2.5 and 4, and is found to be non-corrosive. As it is usually in a solid white powder or flake, it is dissolved in water for use. Sodium benzoate dissociates to form sodium ions and benzoic acid ions. The functional group that is found to be present is both the hydrocarbon derivative, ester, and the hydrocarbon, aromatic benzene ring. The ester is formed when carboxylic acid reacts with alcohol, which is how sodium benzoate is created. Likewise, since it lacks the hydroxide (-OH), it is less soluble in water, less polar, and is not acidic. The properties of this molecule contributes to its uses as described below.
Molecular Formula: NaC7H5O2 | Molar Mass: 144.11 g/mol | Melting Point: 300 °C | Density: 1.50 g/cm³
Sodium benzoate is a white, crystalline powder that is a result of the reaction of benzoic acid with sodium hydroxide. In large concentrations, it holds an unpleasant, saline, astringent taste. The optimum pH of this chemical is between 2.5 and 4, and is found to be non-corrosive. As it is usually in a solid white powder or flake, it is dissolved in water for use. Sodium benzoate dissociates to form sodium ions and benzoic acid ions. The functional group that is found to be present is both the hydrocarbon derivative, ester, and the hydrocarbon, aromatic benzene ring. The ester is formed when carboxylic acid reacts with alcohol, which is how sodium benzoate is created. Likewise, since it lacks the hydroxide (-OH), it is less soluble in water, less polar, and is not acidic. The properties of this molecule contributes to its uses as described below.
|
|
Uses of Sodium Benzoate
In the industry, this organic chemical is used as a preservative in which it is found naturally or synthesized in chemical laboratories. There is a variety of possibilities as to how this substance is put to use in today's society due to it's ubiquitous properties and low costs. Not only is this sodium benzoate present in the food industry, it is also found in products like rocket fuel, fireworks, and personal care products. Products containing this organic chemical needs to be stored in a cool, dry place to ensure it's proper function. |
Food Industry
In foods and pharmaceutical preparations, it is an anti-fungal preservative which extends the product's shelf life. Anti-fungal agents are substances that destroy fungi by suppressing their ability to grow or reproduce. Food preservatives are described to be substances capable of inhibiting, impeding, or incarcerating the process of acidification, fermentation, or other deterioration of foods. This includes foods such as fast food hamburgers, carbonated drinks, salad dressings, pickles, jams, marmalades, and certain vegetable products.
In foods and pharmaceutical preparations, it is an anti-fungal preservative which extends the product's shelf life. Anti-fungal agents are substances that destroy fungi by suppressing their ability to grow or reproduce. Food preservatives are described to be substances capable of inhibiting, impeding, or incarcerating the process of acidification, fermentation, or other deterioration of foods. This includes foods such as fast food hamburgers, carbonated drinks, salad dressings, pickles, jams, marmalades, and certain vegetable products.
Healthcare Industry
Sodium benzoate is used in a treatment for urea cycle disorders and testing for the liver function. A urea cycle disorder is caused by a mutation that results in a deficiency in an enzyme in the urea cycle. Ultimately, nitrogen is removed from the blood and is converted into urea, which is transferred into urine and excreted from the body. In this genetic disorder, the nitrogen accumulates, leading to the highly toxic substance, ammonia, to build up. This chemical reaches the brain, causing irreversible brain damage, coma, or death. This is where sodium benzoate comes into play. This organic chemical is an ammonia scavenger, in which it helps prevent hyperammonemia by removing ammonia through the usage of alternative pathways.
To test for liver function, more specifically, the detoxifying capacity of one's liver, is through the use of sodium benzoate. Hippuric acid is a substance that is synthesized from sodium benzoate and other bodily functions. Depending on the amount of hippuric acid excreted, medical examiners can determine if any abnormalities are present. The sample process of this is to dilute 6g of sodium benzoate in 250mL of water to be given orally to patients after a light breakfast. After approximately 4 hours later, if more than 4.5g hippuric acid (60% of sodium benzoate) is released in their urine, the patient is normal. If less than 3g of hippuric acid is excreted, it indicates a hepatic disfunction. Sodium benzoate is a substance that is beneficial when used correctly in the healthcare industry.
Sodium benzoate is used in a treatment for urea cycle disorders and testing for the liver function. A urea cycle disorder is caused by a mutation that results in a deficiency in an enzyme in the urea cycle. Ultimately, nitrogen is removed from the blood and is converted into urea, which is transferred into urine and excreted from the body. In this genetic disorder, the nitrogen accumulates, leading to the highly toxic substance, ammonia, to build up. This chemical reaches the brain, causing irreversible brain damage, coma, or death. This is where sodium benzoate comes into play. This organic chemical is an ammonia scavenger, in which it helps prevent hyperammonemia by removing ammonia through the usage of alternative pathways.
To test for liver function, more specifically, the detoxifying capacity of one's liver, is through the use of sodium benzoate. Hippuric acid is a substance that is synthesized from sodium benzoate and other bodily functions. Depending on the amount of hippuric acid excreted, medical examiners can determine if any abnormalities are present. The sample process of this is to dilute 6g of sodium benzoate in 250mL of water to be given orally to patients after a light breakfast. After approximately 4 hours later, if more than 4.5g hippuric acid (60% of sodium benzoate) is released in their urine, the patient is normal. If less than 3g of hippuric acid is excreted, it indicates a hepatic disfunction. Sodium benzoate is a substance that is beneficial when used correctly in the healthcare industry.
Fuel Industry
This organic chemical is used to produce rocket fuel and is found in fireworks as a fuel in whistle mix. In rocket fuel, sodium benzoate is usually used in place of potassium benzoate. Both rocket fuel and fireworks require whistle mix, which is a fast burning propellant. It is only suitable for amateur rockets and generally small in size such as Saturn missiles and Bottle-rockets. A sample mix contains 70% potassium chlorate and 30% sodium benzoate. Advantages to this fuel is that it has a very fast burn rate; however, that is also a disadvantage because it is too fast and hard to make large reliable motors with its instability. In positive ways, this chemical is easy to purchase and cheaper than potassium benzoate, but cautions must be kept in mind when using this product. Although this substance has limited use in the fuel industry, it is evidently still used, and is a chemical many individuals consume on a daily basis.
This organic chemical is used to produce rocket fuel and is found in fireworks as a fuel in whistle mix. In rocket fuel, sodium benzoate is usually used in place of potassium benzoate. Both rocket fuel and fireworks require whistle mix, which is a fast burning propellant. It is only suitable for amateur rockets and generally small in size such as Saturn missiles and Bottle-rockets. A sample mix contains 70% potassium chlorate and 30% sodium benzoate. Advantages to this fuel is that it has a very fast burn rate; however, that is also a disadvantage because it is too fast and hard to make large reliable motors with its instability. In positive ways, this chemical is easy to purchase and cheaper than potassium benzoate, but cautions must be kept in mind when using this product. Although this substance has limited use in the fuel industry, it is evidently still used, and is a chemical many individuals consume on a daily basis.
Personal Care & Cosmetics Industry
In this industry, it may include anti-aging creams, toothpaste, dentifrice cleaners, shampoo, and mouthwashes. In low concentrations, these products lower the safety concerns. However, in different countries, different laws state the amount of sodium benzoate that is legal to add to products. Through import and export, this may raise concerns to consumers. In general, there is still an uncertainty with the safety of the use of this substance in this industry. There has not been a sufficient amount of tests preformed to determine the safety of this chemical in the use of the personal care and cosmetics industry. Usage rates for this industry is usually limited to a maximum of 1%.
In this industry, it may include anti-aging creams, toothpaste, dentifrice cleaners, shampoo, and mouthwashes. In low concentrations, these products lower the safety concerns. However, in different countries, different laws state the amount of sodium benzoate that is legal to add to products. Through import and export, this may raise concerns to consumers. In general, there is still an uncertainty with the safety of the use of this substance in this industry. There has not been a sufficient amount of tests preformed to determine the safety of this chemical in the use of the personal care and cosmetics industry. Usage rates for this industry is usually limited to a maximum of 1%.
Other Industries
Surprisingly, this addictive has also made itself into tobacco and water-based paint. Sodium Benzoate acts as a preservative or corrosion inhibitor for the paint. This organic chemical does not have direct relationship towards the negative impact it poses towards the environment and human health in these products, but since it is combined with different products and the means of usage, it does.
Surprisingly, this addictive has also made itself into tobacco and water-based paint. Sodium Benzoate acts as a preservative or corrosion inhibitor for the paint. This organic chemical does not have direct relationship towards the negative impact it poses towards the environment and human health in these products, but since it is combined with different products and the means of usage, it does.
